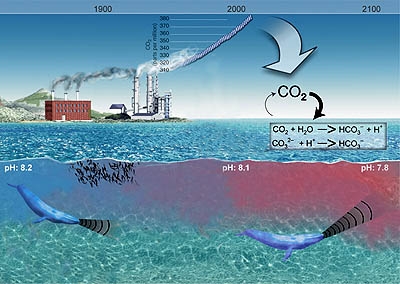
While the livelihoods of more than three billion people depend on oceanic resources, the ocean also provides a large fraction of the oxygen we breathe and absorbs greenhouse gases, mitigating their effects in the atmosphere. Playing a key role in the Earth’s climate and weather systems, as well as in the global carbon cycle, the ocean is an immeasurable force of nature. However, human activities have fundamentally altered the ocean’s chemical composition. Since the late 1980s, 95 per cent of open ocean surface water has become more acidic. Oceans absorb about 30 per cent of carbon dioxide (CO2) we produce, reducing the pH of seawater. This process is known as ocean acidification. With atmospheric CO2 levels 50 per cent above pre-industrial levels, the problem is getting worse.
What is pH and acidity?
pH is the measure of the acidity or basicity of a liquid solution. A solution’s pH represents the concentration of hydrogen ions (H+) and hydroxyl ions (OH-) on a scale of 0 to 14. Pure water has a pH of 7 and is neutral – neither acidic nor basic – with equal concentrations of H+ and OH?. A solution with a pH lower than 7 is acidic, while a solution with a pH greater than 7 is basic. The pH scale is logarithmic, so a decrease of one pH unit is a ten-fold increase in acidity.
The ocean is slightly basic. Prior to the Industrial Revolution of the 18th to 19th centuries, the ocean’s average pH was about 8.2. Today, the ocean’s average pH is 8.1. This means that the ocean today is about 30 per cent more acidic then in pre-industrial times. By 2100, the pH of the ocean could decrease to about 7.8, making the ocean 150 percent more acidic and affecting half of all marine life, according to the Intergovernmental Panel on Climate Change (IPCC) Sixth Assessment Report.
What is the effect of a more acidic ocean?
Ocean acidification threatens marine ecosystems, which also affects populations who rely on the ocean as a source of income and diet. Over three billion people depend on marine and coastal biodiversity for their livelihoods.
For marine ecosystems, ocean acidification presents a two-fold challenge: higher acidity and lower availability of carbonate ions (CO32-). Calcifying organisms – such as oysters, crabs, sea urchins, lobsters and coral – need CO32- to build and maintain their shells and skeletons. Furthermore, studies suggest marine shells and skeletons may dissolve more easily as pH decreases. Scientists are studying the extent to which calcifying organisms are affected by acidification and how some organisms may be more sensitive than others.
Energy spent by marine organisms overcoming more acidic conditions may reduce the energy available for physiological processes, such as reproduction and growth, threatening the stability of food chains that would affect the ecosystem resilience and economic activities, such as fisheries and tourism.
Credit : International atomic energy agency
Picture Credit : Google